TL;DR: CJC-1295 DAC vs no DAC is one of the most studied comparisons in GHRH analog peptide research. The two variants share the same core GHRH receptor agonist sequence but differ fundamentally in pharmacokinetics: the no-DAC form (also called Modified GRF 1-29) has an approximate half-life of 30 minutes and produces pulsatile GH release, while the DAC form uses albumin-binding chemistry to extend half-life to approximately 6-8 days, producing sustained GH and IGF-1 elevation in published research models. This guide covers Drug Affinity Complex technology, albumin binding mechanisms, and the implications of each profile for research protocol design.
Table of Contents
- What Is CJC-1295 and How Does It Relate to GHRH?
- What Is the Drug Affinity Complex (DAC) and How Does It Work?
- CJC-1295 DAC vs No DAC: How Do Half-Lives Compare in Research Models?
- How Do GH Pulse Patterns Differ Between CJC-1295 DAC and No DAC?
- What Does Published Research Show About CJC-1295 DAC and IGF-1?
- CJC-1295 DAC vs No DAC: Side-by-Side Research Comparison Table
- What Are the Protocol Design Implications for Laboratory Research?
- Frequently Asked Questions
- References
What Is CJC-1295 and How Does It Relate to GHRH?
CJC-1295 is a synthetic analog of Growth Hormone-Releasing Hormone (GHRH), the endogenous 44-amino acid hypothalamic peptide that stimulates GH secretion from the anterior pituitary via the GHRH receptor. Native GHRH(1-29) – the minimal bioactive fragment – is rapidly degraded in vivo by dipeptidyl peptidase IV (DPP-IV) and other proteases, limiting its research utility for studies requiring sustained GHRH receptor stimulation. [4]
CJC-1295 was developed as a stabilized GHRH analog incorporating four amino acid substitutions that confer resistance to enzymatic degradation. These modifications – documented in Jetté and colleagues’ identification paper – allow the base peptide sequence to retain GHRH receptor agonist activity with meaningfully improved stability compared to native GHRH(1-29). [5]
The term “CJC-1295” has become a source of nomenclature confusion in research contexts because it is used both for the DAC-bearing compound (the original CJC-1295) and colloquially for the DAC-free stabilized sequence (more accurately designated Modified GRF 1-29 or Mod-GRF). Understanding the CJC-1295 DAC vs no DAC distinction is therefore important for accurately interpreting the published literature and designing in vitro research protocols based on specific pharmacokinetic profiles.
Scientific literature indicates that GHRH analogs represent an important class of synthetic peptide tools for studying the GH axis in preclinical research models, with applications ranging from receptor binding characterization to GH secretion dynamics. [4] [5]
What Is the Drug Affinity Complex (DAC) and How Does It Work?
The Drug Affinity Complex (DAC) is a reactive chemical modification – specifically an N-hydroxysuccinimide (NHS) ester group – appended to the CJC-1295 peptide sequence. The NHS-ester group is designed to form a covalent bond with the lysine residues on endogenous albumin following introduction into a biological system. [5]
The albumin-binding strategy as a general approach for improving peptide and protein pharmacokinetics was characterized by Dennis and colleagues, who documented that covalent or high-affinity binding to albumin – the most abundant plasma protein, with a half-life of approximately 19 days – dramatically extends the circulating half-life of attached molecules. [3] The rationale is straightforward: albumin’s size (approximately 67 kDa) prevents renal filtration, and albumin itself is subject to FcRn-mediated recycling that protects it from degradation.
When CJC-1295 with DAC binds albumin, the resulting conjugate retains GHRH receptor agonist activity while benefiting from albumin’s extended plasma persistence. [5] Jetté and colleagues confirmed that CJC-1295 albumin bioconjugates activate the GRF receptor on the anterior pituitary in rat models, validating that the albumin-bound form retains biological activity at the target receptor. [5]
The no-DAC form (Modified GRF 1-29) lacks this reactive group entirely. Without albumin binding, clearance follows standard peptide kinetics – primarily through renal filtration and proteolytic degradation – resulting in the substantially shorter half-life profile characteristic of this variant.
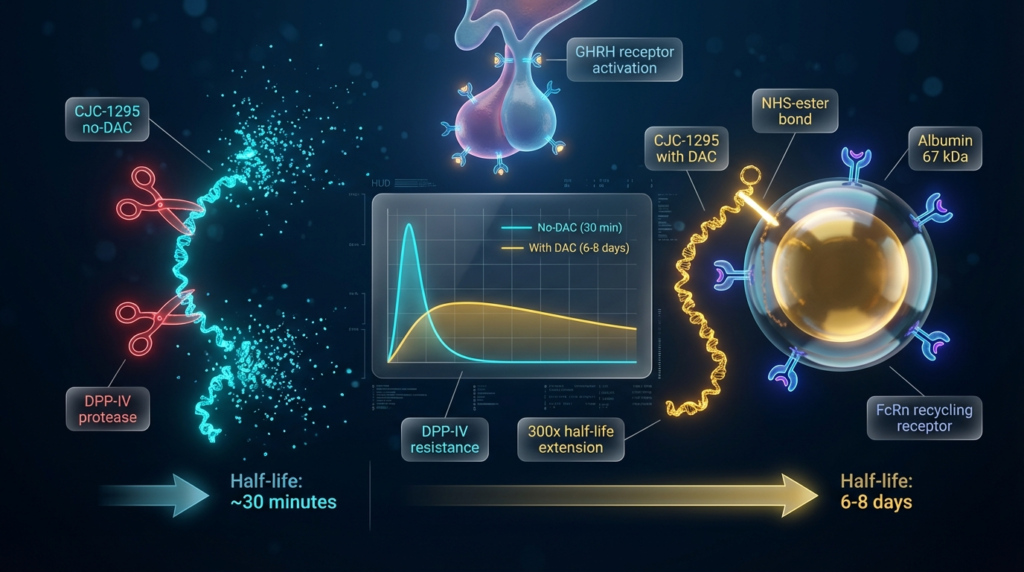
CJC-1295 DAC vs No DAC: How Do Half-Lives Compare in Research Models?
The half-life difference between CJC-1295 DAC and no DAC represents the most practically significant distinction for research protocol design. Published data characterize the two profiles clearly:
CJC-1295 No DAC (Modified GRF 1-29): The stabilized GHRH analog without albumin binding has an approximate half-life of 30 minutes in vivo models. This profile is consistent with other mid-length synthetic peptides that benefit from proteolytic resistance modifications but lack a plasma protein binding mechanism. The short half-life produces a defined, time-limited GHRH receptor stimulation window – useful for research designs requiring acute, pulsatile GH stimulation within a controlled experimental timeframe.
CJC-1295 with DAC: Published clinical research by Teichman and colleagues documented that a single administration of CJC-1295 DAC produced elevated GH and IGF-1 levels for periods extending beyond one week in healthy subjects. [1] This extended activity profile is directly attributable to the albumin-binding mechanism. [3] [5] Half-life estimates from published data place CJC-1295 DAC in the range of 6-8 days – a pharmacokinetic profile that is exceptional among synthetic peptides and substantially changes the parameters of any research protocol incorporating this compound.
This half-life difference – roughly 300-fold between the two variants – is not a minor pharmacological nuance. It defines fundamentally different research tool categories: one for acute stimulation studies and one for sustained, extended-duration investigation of the GH-IGF-1 axis.
How Do GH Pulse Patterns Differ Between CJC-1295 DAC and No DAC?
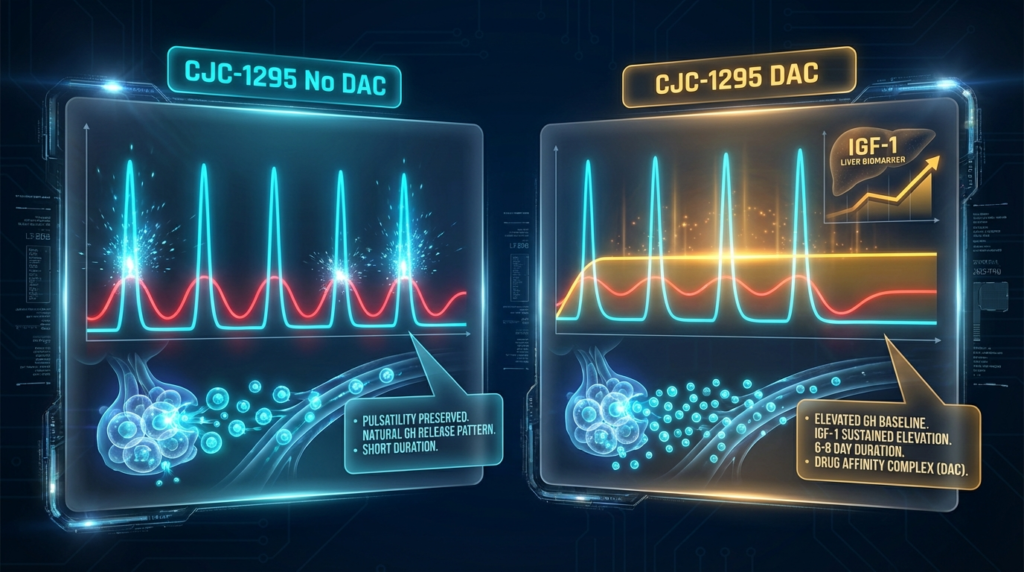
The pulsatility of GH secretion is a biologically important parameter. Endogenous GH is released in discrete pulses from the anterior pituitary, with pulse frequency and amplitude regulated by the interplay of GHRH (stimulatory) and somatostatin (inhibitory) inputs from the hypothalamus. This pulsatile architecture has downstream signaling implications – certain GH-dependent processes respond differently to pulsatile vs continuous GH exposure in preclinical research models.
CJC-1295 No DAC: The short half-life of the no-DAC form means that GHRH receptor stimulation is time-limited, allowing somatostatin’s inhibitory tone to reassert between stimulation events. The resulting GH secretion pattern in research models mirrors the pulsatile profile of endogenous GH release, making Modified GRF 1-29 a tool of choice for studies that aim to preserve or study discrete GH pulse dynamics.
CJC-1295 with DAC: Ionescu and Frohman published a study specifically examining whether pulsatile GH secretion persists during continuous CJC-1295 stimulation. Their findings documented that pulsatile GH secretion does persist even under the sustained GHRH receptor stimulation provided by CJC-1295 DAC – though the baseline GH levels are elevated compared to unstimulated conditions. [2] This finding is significant for research protocol design because it indicates that the extended half-life of CJC-1295 DAC does not abolish GH pulsatility, but rather elevates the overall GH secretory baseline while preserving pulse architecture.
The distinction between elevated-baseline pulsatile GH and true continuous GH exposure has implications for interpreting downstream IGF-1 and GH-responsive biomarker data in preclinical studies.
What Does Published Research Show About CJC-1295 DAC and IGF-1?
IGF-1 (Insulin-like Growth Factor-1), produced primarily by the liver in response to GH signaling, is a standard downstream biomarker used in GH axis research. Its extended half-life relative to GH itself (hours vs minutes) makes it a more stable index of GH secretory status in research models.
Teichman and colleagues published clinical data showing that CJC-1295 DAC produced dose-dependent, sustained increases in both GH and IGF-1 that persisted for the duration of the observation period – up to 28 days in some groups. [1] The authors documented a mean half-life for CJC-1295 DAC of approximately 6-8 days across the dose groups studied, with IGF-1 elevations tracking the extended GH stimulation profile. [1]
These published findings established CJC-1295 DAC as a research tool capable of sustained GH axis stimulation over timeframes not achievable with shorter-acting GHRH analogs, making it relevant for preclinical studies examining chronic GH-IGF-1 axis effects. [1] [4]
CJC-1295 DAC vs No DAC: Side-by-Side Research Comparison Table
| Property | CJC-1295 No DAC (Modified GRF 1-29) | CJC-1295 with DAC |
|---|---|---|
| Common Research Name | Modified GRF(1-29) | CJC-1295 |
| DAC Technology | No | Yes – NHS-ester albumin binding [5] |
| Albumin Binding | No | Covalent – via NHS-ester group [3] |
| Approximate Half-life | ~30 minutes | ~6-8 days [1] |
| Receptor Target | GHRH receptor | GHRH receptor (via albumin carrier) [5] |
| GH Release Pattern (Research Models) | Pulsatile – short stimulation window | Sustained elevation with preserved pulsatility [2] |
| IGF-1 Effects (Published Data) | Short-term elevation | Sustained multi-day elevation [1] |
| Research Application | Acute, short-window GH axis studies | Extended-duration GH axis studies |
| Proteolytic Stability | Improved vs native GHRH(1-29) | Improved – plus albumin protection [5] |
| RUO Classification | Yes – in vitro research only | Yes – in vitro research only |
What Are the Protocol Design Implications for Laboratory Research?
For researchers designing in vitro or preclinical studies around GHRH axis pharmacology, the CJC-1295 DAC vs no DAC comparison has direct methodological implications.
Short-duration, pulsatile stimulation studies: Modified GRF 1-29 (no DAC) is appropriate for research designs requiring a defined, time-limited GHRH receptor activation event. Its ~30-minute half-life allows researchers to establish clear stimulation and washout windows, making it suitable for acute signaling studies, receptor activation kinetics experiments, and protocols where pulsatile GH release is the target variable.
Extended-duration GH axis studies: CJC-1295 with DAC is the appropriate research tool when sustained GH and IGF-1 elevation over days is the experimental requirement. Published data confirm that a single administration can maintain elevated GH axis activity for up to one week, enabling researchers to study sustained GHRH receptor stimulation effects without repeated dosing events. [1] [2]
Combination protocol research: Scientific literature documents the co-administration of CJC-1295 DAC with GHS-R1a agonists (such as ipamorelin and GHRP-6) in preclinical research settings. The mechanistic rationale is that GHRH receptor activation (via CJC-1295) and GHS-R1a receptor activation act through complementary, synergistic pathways to amplify GH pulse amplitude.
For a broader framework of current research protocols across the synthetic peptide field, the Molecular Edge 2026 Master Index of Peptide Research Protocols serves as a reference hub.
Frequently Asked Questions
What is the difference between CJC-1295 DAC and CJC-1295 no DAC?
The core difference is pharmacokinetic. Both variants share a stabilized GHRH receptor agonist sequence, but CJC-1295 with DAC incorporates an NHS-ester reactive group that forms a covalent bond with endogenous albumin, extending its half-life to approximately 6-8 days. [1] [5] The no-DAC form (Modified GRF 1-29) lacks this group and clears within approximately 30 minutes, producing short-duration, pulsatile GHRH receptor stimulation.
What does DAC stand for in CJC-1295 DAC?
DAC stands for Drug Affinity Complex – a reactive NHS-ester chemical modification appended to the peptide sequence that enables covalent albumin binding after introduction into a biological system. [5] The albumin-binding mechanism is the basis for CJC-1295 DAC’s extended pharmacokinetic profile. [3]
Does pulsatile GH secretion persist with CJC-1295 DAC?
Published research by Ionescu and Frohman specifically examined this question and documented that pulsatile GH secretion persists during continuous CJC-1295 stimulation, with GH pulses occurring against an elevated baseline. [2] This finding distinguishes CJC-1295 DAC’s effect from true continuous, non-pulsatile GH exposure.
What is Modified GRF 1-29?
Modified GRF 1-29 is the common research name for CJC-1295 without the DAC modification – the stabilized GHRH(1-29) analog with four amino acid substitutions for proteolytic resistance, without the albumin-binding NHS-ester group. It is used in research as the short-acting GHRH analog for acute GH axis stimulation studies. Both Modified GRF 1-29 and CJC-1295 DAC are classified as Research Use Only (RUO) compounds.
What IGF-1 effects has CJC-1295 DAC shown in published research?
Teichman and colleagues documented that CJC-1295 DAC produced sustained, dose-dependent IGF-1 elevation that persisted for up to 28 days in some groups – tracking the extended GH stimulation profile conferred by the albumin-binding mechanism. [1] These findings derive from a published clinical study; all data are specific to the research context and do not support personal use conclusions.
Are CJC-1295 DAC and no DAC approved for human use?
No. Both CJC-1295 with DAC and CJC-1295 without DAC (Modified GRF 1-29) carry a Research Use Only (RUO) classification and are not approved by the FDA or any regulatory body for human or animal consumption, or for any therapeutic or diagnostic application. Molecular Edge Peptides supplies both variants exclusively as research-grade compounds for controlled in vitro laboratory investigation.
Disclaimer: All products sold by Molecular Edge Peptides are strictly intended for laboratory research use only (in vitro). They are not approved for human or animal consumption, or for any form of therapeutic, diagnostic, or clinical use. The information in this article is for educational and scientific reference purposes only. We do not provide usage instructions, dosing guidelines, or any advice regarding personal application of our products. Always consult relevant regulatory frameworks before conducting research with these compounds.
References
- Teichman SL, Neale A, Lawrence B, Gagnon C, Castaigne JP, Frohman LA. “Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults.” J Clin Endocrinol Metab. 2006;91(3):799-805. https://pubmed.ncbi.nlm.nih.gov/16352683/
- Ionescu M, Frohman LA. “Pulsatile secretion of growth hormone (GH) persists during continuous stimulation by CJC-1295, a long-acting GH-releasing hormone analog.” J Clin Endocrinol Metab. 2006;91(12):4792-4797. https://pubmed.ncbi.nlm.nih.gov/16984980/
- Dennis MS, Zhang M, Meng YG, et al. “Albumin binding as a general strategy for improving the pharmacokinetics of proteins.” J Biol Chem. 2002;277(38):35035-35043. https://pubmed.ncbi.nlm.nih.gov/12117986/
- Smith RG. “Development of growth hormone secretagogues.” Endocr Rev. 2005;26(3):346-360. https://pubmed.ncbi.nlm.nih.gov/15774820/
- Jetté L, Léger R, Thibaudeau K, et al. “Human growth hormone-releasing factor (hGRF)1-29-albumin bioconjugates activate the GRF receptor on the anterior pituitary in rats: identification of CJC-1295 as a long-lasting GRF analog.” Endocrinology. 2005;146(7):3052-3058. https://pubmed.ncbi.nlm.nih.gov/15760874/
- Fosgerau K, Hoffmann T. “Peptide therapeutics: current status and future directions.” Drug Discov Today. 2015;20(1):122-128. https://pubmed.ncbi.nlm.nih.gov/25450771/


