TL;DR: Epithalon (Ala-Glu-Asp-Gly) is a synthetic tetrapeptide derived from a pineal gland peptide fraction. Published epithalon peptide research has examined its relationship to telomerase activity in human somatic cells, aging biomarkers in animal models, and its origins in Khavinson’s peptide bioregulation research program. This guide summarizes the scientific literature on Epithalon’s mechanisms, research history, and documented activity across model systems – for educational and laboratory reference purposes only.
Table of Contents
- What Is Epithalon and What Is Its Chemical Structure?
- What Is the Research History Behind Epithalon Peptide Research?
- How Does Epithalon Interact with Telomerase in Published Studies?
- What Is the Pineal Gland Connection in Epithalon Research?
- What Aging Biomarkers Have Been Studied Alongside Epithalon in Animal Models?
- Epithalon Research Summary: Published Model Findings at a Glance
- Frequently Asked Questions
- References
What Is Epithalon and What Is Its Chemical Structure?
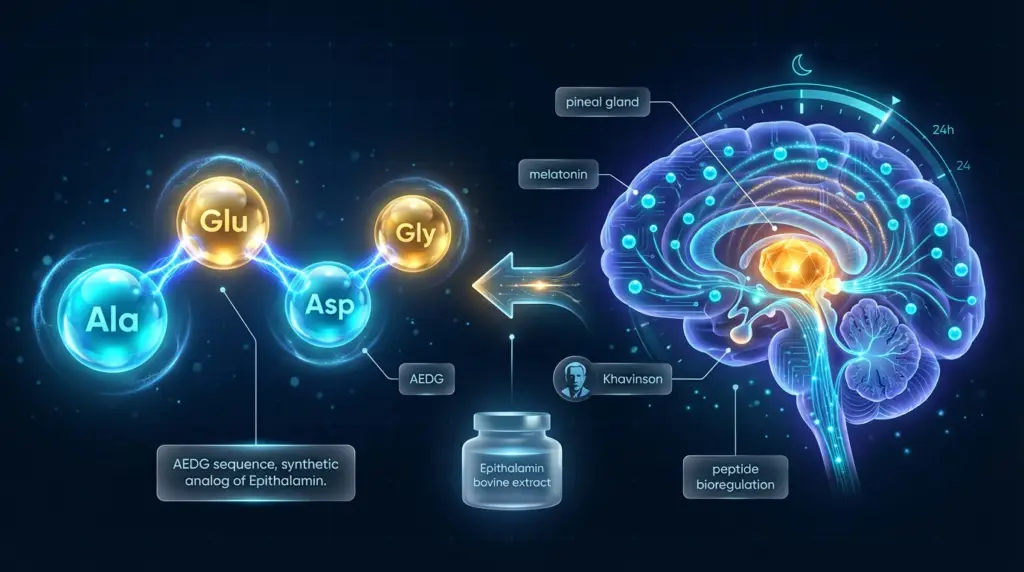
Epithalon peptide research centers on a synthetic tetrapeptide composed of four amino acid residues in the sequence Alanine-Glutamic acid-Aspartic acid-Glycine (Ala-Glu-Asp-Gly, or AEDG). As a tetrapeptide, Epithalon is among the smallest synthetic peptides studied in the context of aging and neuroendocrine biology – a structural simplicity that has made it a useful tool in in vitro research for isolating specific molecular interactions.
The compound is a synthetic analog of a bioactive peptide fraction isolated from bovine pineal gland tissue, known as Epithalamin. The synthesis of Epithalon as a defined four-residue sequence was part of a broader research program aimed at identifying the minimal active sequences responsible for the biological activities observed in pineal-derived peptide extracts. [3]
Epithalon’s small molecular size – well below the 500 Dalton threshold relevant in certain absorption research contexts (see the Molecular Edge guide to the 500 Dalton rule and peptide penetration research) – has made it accessible for in vitro cell culture work and in vivo animal model studies. Scientific literature classifies it as a research-grade synthetic peptide, supplied exclusively under a Research Use Only (RUO) classification for controlled laboratory investigation.
What Is the Research History Behind Epithalon Peptide Research?
The scientific foundation of epithalon peptide research is rooted in the work of Vladimir Khavinson and colleagues at the St. Petersburg Institute of Bioregulation and Gerontology. Over several decades, Khavinson’s research program developed the concept of “peptide bioregulation” – the hypothesis that short peptide sequences derived from organ-specific tissues could influence biological processes associated with aging at the cellular level. [3]
Epithalamin, the polypeptide pineal extract from which Epithalon was derived, was among the first compounds studied within this framework. Khavinson and colleagues later synthesized the tetrapeptide AEDG sequence as a chemically defined, reproducible research tool to enable more precise mechanistic investigation than was possible with the complex polypeptide extract. [3]
Published reviews by Anisimov and Khavinson summarize the peptide bioregulation research program across multiple model systems, documenting studies in invertebrates, rodents, and in vitro cell culture spanning from the 1980s through the 2010s. [3] This body of published literature represents the primary scientific record for Epithalon peptide research and forms the reference base for any serious laboratory investigation of this compound.
The research history of Epithalon also reflects broader developments in longevity science – the intersection of telomere biology, neuroendocrinology, and aging biomarker research that has driven growing interest in pineal-related peptide compounds within the scientific community.
How Does Epithalon Interact with Telomerase in Published Studies?
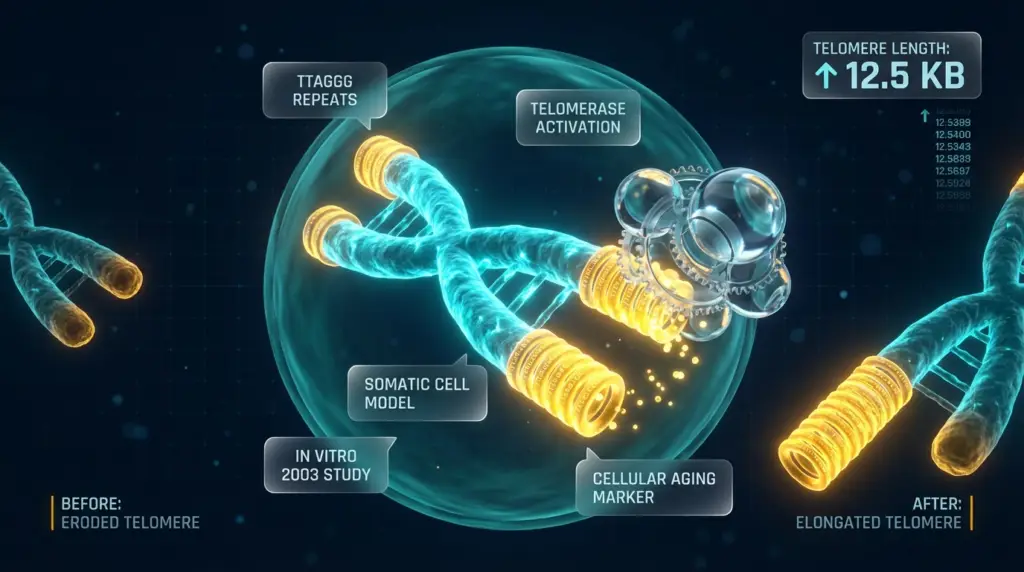
Telomere biology is the most widely cited dimension of epithalon peptide research in the scientific literature. Telomeres are repetitive nucleotide sequences (TTAGGG in humans) that cap the ends of chromosomes, protecting genomic integrity. With each cell division, telomeres progressively shorten – a process associated with cellular senescence. Telomerase, the enzyme responsible for adding telomeric repeats, is expressed at low or absent levels in most adult somatic cells, making its activation a subject of significant interest in aging and cellular biology research.
Khavinson and colleagues published a study in 2003 examining Epithalon’s effect on telomerase activity in human somatic cells in vitro. The study reported that Epithalon induced telomerase activity and was associated with telomere elongation in the examined cell culture system. [1] This finding placed Epithalon within the context of telomerase activation research – a field that had gained substantial momentum following the Nobel Prize-recognized work on telomere biology in 2009.
It is important for researchers to note that the published in vitro telomere findings document observations in controlled cell culture conditions. [1] Extrapolating these results to organismal aging or clinical contexts requires independent validation across additional model systems, and no therapeutic conclusions are supported by this body of published evidence.
Scientific literature also situates Epithalon research within the broader framework of aging biomarker investigation, where telomere length serves as one of several measurable endpoints in studies examining cellular aging processes. [3]
What Is the Pineal Gland Connection in Epithalon Research?
The pineal gland is a small neuroendocrine structure in the brain, most known for its role in melatonin synthesis and circadian rhythm regulation. In the context of aging research, the pineal gland has attracted scientific attention due to evidence from animal studies that pineal function declines with age and that pineal-derived factors may influence neuroendocrine aging processes. [3]
Epithalon’s origin as a synthetic analog of a pineal peptide fraction situates it directly within pineal-neuroendocrine research. Published studies by Khavinson and colleagues have examined the relationship between Epithalon and melatonin secretion patterns in animal models, investigating whether the tetrapeptide interacts with the regulatory networks that govern pineal gland function during aging. [3]
Scientific literature indicates that the neuroendocrine hypothesis underlying Epithalon research proposes that the pineal gland’s peptide secretory activity represents a regulatory input into broader aging biology – and that short synthetic peptides derived from pineal tissue may serve as tools to study these interactions in controlled laboratory settings. [3]
Researchers interested in the neuroendocrine dimension of longevity peptide research can explore the Molecular Edge compound catalog, including blend formulations such as the KLOW Blend, which combines multiple research-grade peptides relevant to this area of investigation.
What Aging Biomarkers Have Been Studied Alongside Epithalon in Animal Models?
Epithalon peptide research in animal models has examined a range of endpoints beyond telomere biology. Published studies document investigations across multiple model organisms and aging-relevant biological endpoints:
Invertebrate lifespan models: Khavinson and colleagues published a study in Drosophila melanogaster documenting lifespan increase in the invertebrate model following Epithalon treatment. [2] While invertebrate lifespan data do not translate directly to mammalian biology, such studies are standard early-stage tools in longevity research for establishing initial biological activity.
Rodent tumor models: Anisimov and colleagues examined Epithalon’s effect in HER-2/neu transgenic mice, a model prone to spontaneous mammary tumor development. The published study documented an inhibitory effect on tumor development in this transgenic model. [5] Separately, Kossoy and colleagues examined Epithalon in a rat colon carcinogenesis model, investigating effects on cell proliferative activity and apoptosis in colon tumors and mucosa. [4]
Oxidative stress markers: Kozina and colleagues published data on Epithalon’s antioxidant properties in vitro, documenting activity against reactive oxygen species in the examined model. [6] Oxidative stress is a well-established biomarker in aging biology research, making antioxidant profiling a standard component of longevity-related peptide investigation.
Epithalon Research Summary: Published Model Findings at a Glance
| Research Model | Biological Endpoint Investigated | Key Observation in Published Studies | Reference |
|---|---|---|---|
| Human somatic cells (in vitro) | Telomerase activity and telomere elongation | Telomerase induction and telomere elongation reported | [1] |
| Drosophila melanogaster (invertebrate) | Lifespan | Lifespan increase documented in invertebrate model | [2] |
| HER-2/neu transgenic mice (rodent) | Mammary tumor development | Inhibitory effect on tumor development reported | [5] |
| Rat colon carcinogenesis model | Cell proliferation and apoptosis in colon tissue | Epithalon examined for effects on proliferative activity | [4] |
| In vitro oxidative stress models | Antioxidant activity | Antioxidant properties documented | [6] |
| Multiple animal models (review) | Aging biomarkers – neuroendocrine parameters | Peptide bioregulation framework summarized across models | [3] |
All findings listed above derive from published preclinical or in vitro research. None of these data support clinical conclusions or personal use recommendations. These compounds are supplied for laboratory research use only.
Frequently Asked Questions
What is Epithalon peptide and what is its amino acid sequence?
Epithalon is a synthetic tetrapeptide with the amino acid sequence Ala-Glu-Asp-Gly (AEDG). It was developed as a defined synthetic analog of Epithalamin, a peptide fraction extracted from bovine pineal gland tissue, as part of Khavinson’s peptide bioregulation research program. [3] It is classified as a Research Use Only (RUO) compound for in vitro laboratory research exclusively.
What has epithalon peptide research shown about telomeres?
Published in vitro research by Khavinson and colleagues documented that Epithalon induced telomerase activity and was associated with telomere elongation in human somatic cells in a cell culture model. [1] These findings are specific to the in vitro system studied and do not support clinical or personal use conclusions.
Is there published research on Epithalon and lifespan?
Yes. Khavinson and colleagues published a study examining Epithalon in Drosophila melanogaster, documenting lifespan increase in that invertebrate model. [2] Anisimov and Khavinson have also reviewed broader aging biomarker research across multiple model organisms in published literature. [3] All published lifespan data derive from preclinical animal models.
What is the connection between Epithalon and the pineal gland?
Epithalon was synthesized as an analog of a bioactive fraction derived from bovine pineal gland tissue (Epithalamin). Published research has examined Epithalon’s interactions with neuroendocrine aging parameters – including pineal-related regulatory networks – in animal models. [3] The pineal gland’s role in melatonin synthesis and circadian biology makes it a subject of ongoing interest in aging research.
Who conducted the primary research on Epithalon?
The primary body of Epithalon peptide research was conducted by Vladimir Khavinson and colleagues at the St. Petersburg Institute of Bioregulation and Gerontology, with collaborators including Anisimov and others, published across peer-reviewed journals from the 1990s through the 2010s. [2] [3] The research program examined multiple synthetic peptides as part of a broader peptide bioregulation framework.
Is Epithalon approved for human use?
No. Epithalon carries a Research Use Only (RUO) classification and is not approved by the FDA or any regulatory body for human or animal consumption, or for any therapeutic or diagnostic application. Molecular Edge Peptides supplies Epithalon exclusively as a research-grade compound for controlled in vitro laboratory investigation.
Disclaimer: All products sold by Molecular Edge Peptides are strictly intended for laboratory research use only (in vitro). They are not approved for human or animal consumption, or for any form of therapeutic, diagnostic, or clinical use. The information in this article is for educational and scientific reference purposes only. We do not provide usage instructions, dosing guidelines, or any advice regarding personal application of our products. Always consult relevant regulatory frameworks before conducting research with these compounds.
References
- Khavinson VK, Bondarev IE, Butyugov AA, Tamara DM. “Epithalon peptide induces telomerase activity and telomere elongation in human somatic cells.” Bull Exp Biol Med. 2003;135(6):590-592. https://pubmed.ncbi.nlm.nih.gov/14513035/
- Khavinson VK, Izmaylov DM, Obukhova LK, Malinin VV. “Effect of Epithalon on the lifespan increase in Drosophila melanogaster.” Mech Ageing Dev. 2000;120(1-3):141-149. https://pubmed.ncbi.nlm.nih.gov/10993928/
- Anisimov VN, Khavinson VK. “Peptide bioregulation of aging: results and prospects.” Biogerontology. 2010;11(2):139-149. https://pubmed.ncbi.nlm.nih.gov/19575298/
- Kossoy G, Zandbank J, Tendler E, et al. “Epitalon and colon carcinogenesis in rats: proliferative activity and apoptosis in colon tumors and mucosa.” Int J Mol Med. 2003;12(4):473-477. https://pubmed.ncbi.nlm.nih.gov/12964025/
- Anisimov VN, Khavinson VK, Alimova IN, et al. “Epithalon inhibits tumor development in mammary gland in HER-2/neu transgenic mice.” Int J Cancer. 2002;101(1):7-10. https://pubmed.ncbi.nlm.nih.gov/12209579/
- Kozina LS, Arutjunyan AV, Khavinson VK. “Antioxidant properties of the tetrapeptide Epitalon.” Bull Exp Biol Med. 2007;144(2):213-215. https://pubmed.ncbi.nlm.nih.gov/18399244/


