TL;DR: Both ipamorelin and GHRP-6 act as GHS-R1a agonists that stimulate pulsatile growth hormone release in preclinical and clinical research models. The defining distinction: ipamorelin demonstrates high GH selectivity with minimal cortisol and prolactin co-stimulation in published studies, while GHRP-6 activates a broader hormonal cascade that includes significant ACTH and cortisol co-stimulation. Researchers select between them based on the hormonal specificity required by a given in vitro or preclinical study design.
Table of Contents
- What Are Ipamorelin and GHRP-6 in Peptide Research?
- How Do Ipamorelin and GHRP-6 Bind the GHS-R1a Receptor?
- How Do Ipamorelin and GHRP-6 Compare? Full Specifications
- What Does Published Research Show About GH Selectivity?
- How Do Cortisol and Prolactin Profiles Differ Between Ipamorelin and GHRP-6?
- What Are the Structural Differences Between Ipamorelin and GHRP-6?
- Why Do Researchers Co-Administer GH Secretagogues with GHRH Analogs?
- Frequently Asked Questions
- References
What Are Ipamorelin and GHRP-6 in Peptide Research?
Ipamorelin and GHRP-6 are both synthetic growth hormone secretagogues (GHS) – Research Use Only (RUO) compounds classified as ghrelin receptor agonists that stimulate growth hormone release from pituitary somatotroph cells in preclinical research models.
Both compounds belong to the GHRP (growth hormone-releasing peptide) class, a family of synthetic molecules that act at the GHS-R1a receptor independently of growth hormone-releasing hormone (GHRH). Their discovery and characterization by Cyril Bowers and colleagues across multiple decades of GHS research established the foundational pharmacological framework for understanding ghrelin receptor biology. [4]
The ipamorelin vs GHRP-6 comparison is among the most studied in the GH secretagogue literature – not because the two compounds share identical profiles, but precisely because they diverge in selectivity, hormonal co-stimulation patterns, and downstream research implications. For laboratories investigating GH axis biology, pituitary receptor pharmacology, or somatotroph cell function, understanding this comparison is foundational.
Both compounds are classified as RUO peptides for controlled in vitro laboratory investigation. For background on the research peptide classification framework – including RUO designation, purity standards, and CoA documentation – the complete research peptide classification guide provides foundational context.
How Do Ipamorelin and GHRP-6 Bind the GHS-R1a Receptor?
Both ipamorelin and GHRP-6 act as agonists at the GHS-R1a receptor – the growth hormone secretagogue receptor type 1a – a G-protein coupled receptor (GPCR) expressed in the pituitary gland and hypothalamus that mediates growth hormone release in response to ghrelin and synthetic GHS ligands.
The GHS-R1a receptor was cloned and characterized by Howard et al. in 1996, establishing it as the endogenous binding site for ghrelin and the primary molecular target of the entire GHRP compound class. [3] Activation of GHS-R1a triggers intracellular calcium mobilization via Gq/11-mediated phospholipase C signaling, leading to exocytosis of GH-containing secretory granules from pituitary somatotroph cells.
Both ipamorelin and GHRP-6 bind this receptor with high affinity, but their structural differences produce distinct receptor activation profiles and downstream hormonal consequences. Ipamorelin’s structural features allow it to engage GHS-R1a with high specificity for GH-related pathways, without activating co-stimulatory ACTH and cortisol responses. [1] GHRP-6, by contrast, activates additional receptor-mediated pathways that extend the hormonal response beyond pure somatotroph stimulation.
This receptor-level divergence is the mechanistic foundation of the entire ipamorelin vs GHRP-6 research comparison.
How Do Ipamorelin and GHRP-6 Compare? Full Specifications
| Parameter | Ipamorelin | GHRP-6 |
|---|---|---|
| Peptide class | Pentapeptide | Hexapeptide |
| Amino acid sequence | Aib-His-D-2-Nal-D-Phe-Lys-NH2 | His-D-Trp-Ala-Trp-D-Phe-Lys-NH2 |
| Molecular weight | 711.9 Da | 873.0 Da |
| Primary receptor target | GHS-R1a | GHS-R1a |
| GH stimulation | Yes | Yes |
| Cortisol / ACTH co-stimulation | Minimal in published studies | Significant elevation documented |
| Prolactin co-stimulation | Minimal | Moderate |
| GH selectivity profile | High – GH-selective | Broad hormonal cascade |
| Half-life (approximate) | ~2 hours | ~1-2 hours |
| DPP-IV susceptibility | Partial resistance (D-amino acids) | Limited resistance |
| GHRH synergy in research | Yes | Yes |
| First characterized | Raun et al., 1998 | Bowers et al., 1984 |
| Research classification | RUO – in vitro only | RUO – in vitro only |
Molecular Edge Peptides supplies ipamorelin for laboratory research manufactured to >99% purity with full Certificate of Analysis documentation – HPLC chromatogram and mass spectrometry data included – for qualified in vitro laboratory use exclusively.
What Does Published Research Show About GH Selectivity?
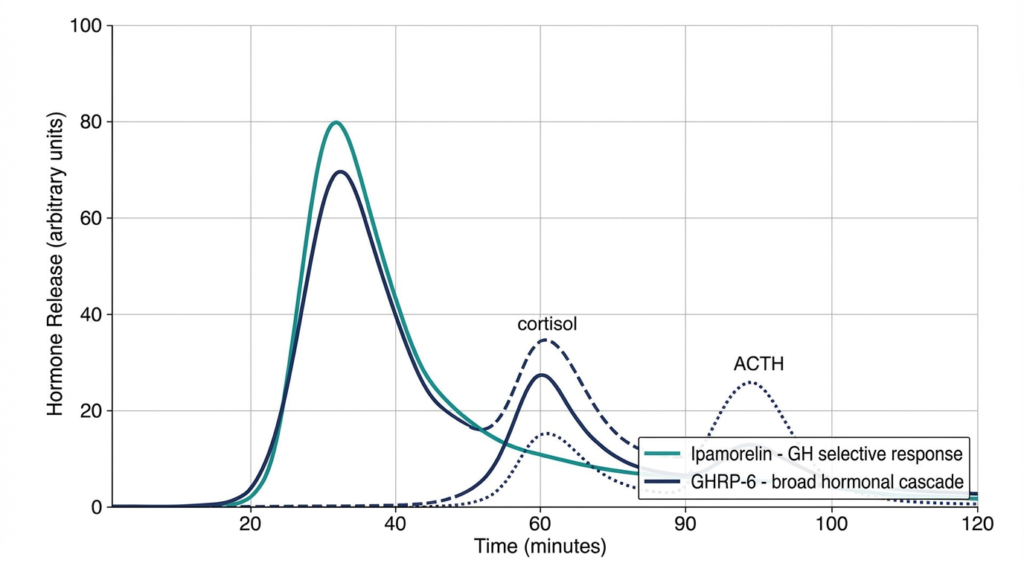
The Raun et al. (1998) study in the European Journal of Endocrinology established ipamorelin as the first GH secretagogue to demonstrate high GH selectivity – stimulating GH release without significant co-stimulation of ACTH, cortisol, or prolactin in preclinical and early human research models. [1]
This selectivity profile distinguishes ipamorelin from all other GHRP compounds characterized up to that point. Prior GHRPs, including GHRP-6 and GHRP-2, consistently triggered corticotroph and lactotroph activation alongside somatotroph stimulation – a pattern documented across multiple clinical research settings. Ipamorelin’s clean GH-selective profile was a pharmacologically novel result within the entire GHS compound class at the time of publication. [1]
For research applications requiring isolated investigation of GH axis biology – without the confounding variable of concurrent cortisol and ACTH changes – ipamorelin’s selectivity profile is a key experimental differentiator. In study designs where the cortisol/ACTH axis is precisely the subject under investigation, GHRP-6’s broader activation pattern makes it the more informative probe for that specific research question.
The literature on GHS selectivity demonstrates a recurring observation: receptor affinity alone does not predict hormonal cascade breadth. Two compounds can bind GHS-R1a with similar affinity while producing distinctly different downstream hormonal profiles – a distinction with real consequences for experimental interpretation and results validity.
How Do Cortisol and Prolactin Profiles Differ Between Ipamorelin and GHRP-6?
GHRP-6 administration in human research subjects produced significant ACTH and cortisol elevation alongside GH release, confirming that GHRP-6 activates hypothalamic-pituitary-adrenal (HPA) axis pathways beyond pure somatotroph stimulation.
The Frieboes et al. (1995) neuroendocrinology study examined the full neuroendocrine effects of GHRP-6 in healthy male subjects and documented not only robust GH secretion but concurrent increases in ACTH and cortisol, with moderate prolactin elevation. [5] This multi-axis hormonal activation reflects GHRP-6’s engagement of receptor-mediated pathways beyond GHS-R1a somatotroph signaling alone.
Ipamorelin showed no significant ACTH, cortisol, or aldosterone changes at doses producing maximal GH secretion across the Raun et al. study’s dose-response analysis, and this selectivity held across a wide dose range – suggesting it is an intrinsic property of ipamorelin’s receptor interaction rather than a dose-dependent artifact. [1]
The Arvat et al. (2001) comparative study further contextualized these profiles by examining the endocrine activity of ghrelin alongside hexarelin and other synthetic secretagogues, confirming that hormonal cascade breadth varies substantially across the GHS compound class and cannot be predicted from GHS-R1a binding affinity alone. [6]
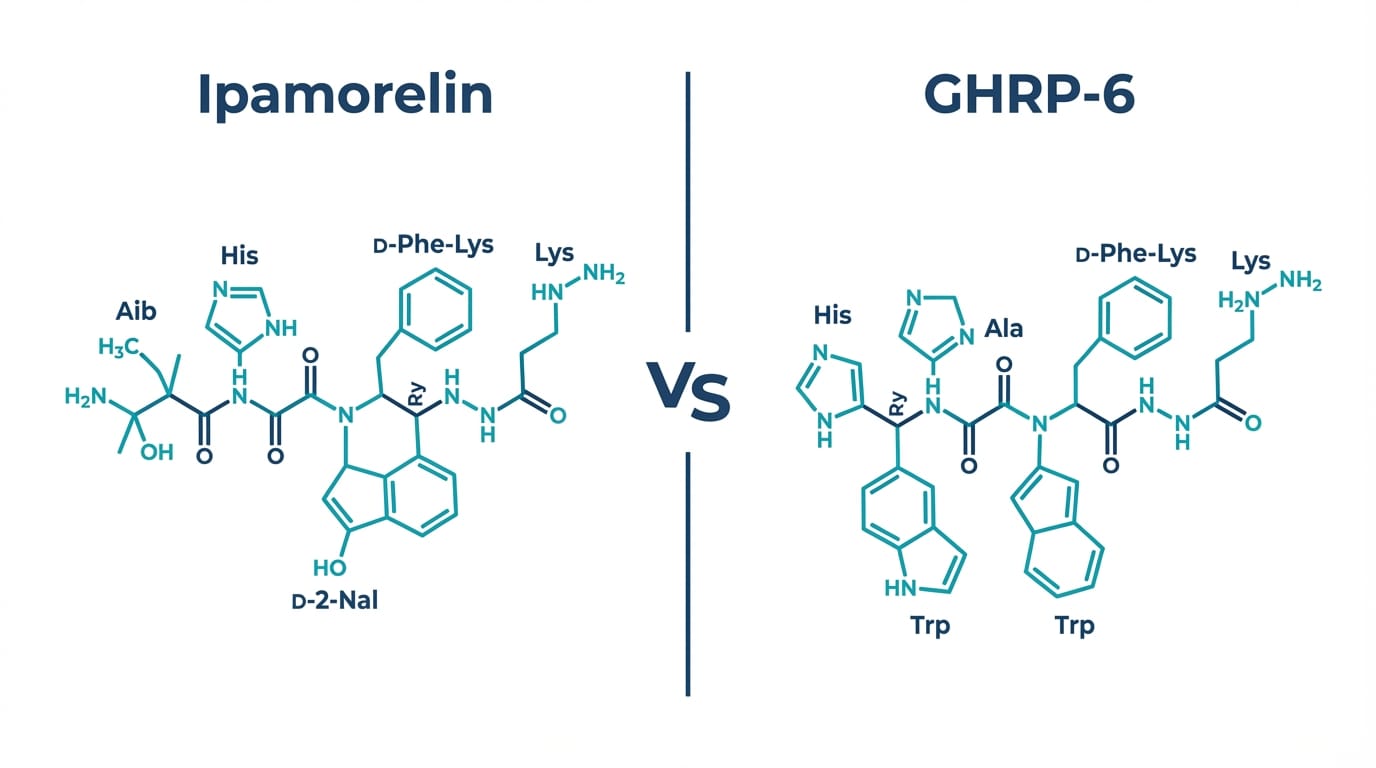
What Are the Structural Differences Between Ipamorelin and GHRP-6?
Ipamorelin is a pentapeptide (5 residues) with the sequence Aib-His-D-2-Nal-D-Phe-Lys-NH2, while GHRP-6 is a hexapeptide (6 residues) with the sequence His-D-Trp-Ala-Trp-D-Phe-Lys-NH2 – two structurally related but functionally distinct GHS-R1a ligands first characterized by Bowers et al. [2]
Key structural distinctions that underlie their functional differences:
- N-terminal residue: Ipamorelin uses alpha-aminoisobutyric acid (Aib) at position 1, a non-proteinogenic alpha-methyl amino acid that confers conformational constraint. GHRP-6 opens with His at position 1.
- Position 3 aromatic residue: Ipamorelin incorporates D-2-naphthylalanine (D-2-Nal), a bulky D-amino acid. GHRP-6 uses D-Trp in the equivalent position. Both residues are critical for GHS-R1a binding affinity, and their structural similarity explains the shared receptor target despite divergent downstream profiles.
- D-amino acid content: Both compounds incorporate D-amino acids at key positions, conferring partial resistance to dipeptidyl peptidase IV (DPP-IV) cleavage compared to all-L-amino acid peptide sequences.
- C-terminal modification: Both are C-terminally amidated (-NH2), a modification that improves metabolic stability and is standard in synthetic research peptide design.
The structural convergence at the key aromatic position likely underlies shared GHS-R1a affinity, while divergence at the N-terminus and chain length contribute to ipamorelin’s selective receptor engagement profile relative to GHRP-6.
Why Do Researchers Co-Administer GH Secretagogues with GHRH Analogs?
GH secretagogues like ipamorelin and GHRP-6 act synergistically with GHRH at the pituitary level – the two mechanistically distinct GH-release pathways potentiate each other, producing greater GH secretion in combination than either class achieves independently in preclinical research models.
This synergy reflects the complementary nature of the two stimulatory mechanisms: GHRH acts via the GHRH-R receptor to increase intracellular cAMP, while GH secretagogues act via GHS-R1a to mobilize intracellular calcium. The two signaling cascades converge at the level of GH secretory granule exocytosis, where their combined effect exceeds either pathway’s independent contribution. [4]
In published research, GHRP compounds are frequently co-administered with GHRH analogs – including modified GRF 1-29 and its albumin-binding derivatives – to investigate combined GH axis stimulation. Ipamorelin’s selective profile makes it a particularly useful co-administration partner when investigators want to isolate the GH-axis contribution of the GHRH analog without cortisol confound from the secretagogue component.
For laboratories studying GHRH analog compounds alongside GH secretagogues, the CJC-1295 DAC and No DAC half-life and GH pulse research comparison covers the mechanistic differences between albumin-binding and standard GHRH analog forms – directly relevant to co-administration study design. Molecular Edge Peptides supplies research-grade CJC-1295 No DAC manufactured to >99% purity with full CoA documentation for qualified in vitro laboratory use.
Frequently Asked Questions
What is the difference between ipamorelin and GHRP-6?
Both ipamorelin and GHRP-6 are GHS-R1a agonists that stimulate growth hormone release in preclinical research models. The primary difference is selectivity: ipamorelin produces GH release with minimal cortisol, ACTH, or prolactin co-stimulation in published studies, while GHRP-6 activates a broader hormonal cascade including significant ACTH and cortisol elevation. Structurally, ipamorelin is a pentapeptide with Aib at the N-terminus, and GHRP-6 is a hexapeptide with His at the N-terminus. Both are RUO-classified compounds for in vitro laboratory research only.
Does ipamorelin raise cortisol in research models?
Raun et al. (1998) found no significant cortisol, ACTH, or aldosterone changes at ipamorelin doses producing maximal GH secretion in preclinical models, establishing ipamorelin as the first GH-selective secretagogue in the published literature. This distinguishes ipamorelin from GHRP-6 and GHRP-2, which produce documented ACTH/cortisol co-stimulation across multiple research studies. All research involving these compounds is conducted under RUO classification for qualified in vitro laboratory use only.
Does GHRP-6 raise cortisol in research?
Yes – the Frieboes et al. (1995) study in Neuroendocrinology documented significant ACTH and cortisol elevation following GHRP-6 administration in healthy research subjects, alongside robust GH secretion. This multi-axis hormonal activation is a consistent finding in GHRP-6 research and distinguishes it from ipamorelin’s GH-selective profile. Researchers studying pure GH axis biology typically select ipamorelin to avoid this cortisol and ACTH confound in their experimental designs.
What receptor do ipamorelin and GHRP-6 act on?
Both compounds act as agonists at the GHS-R1a receptor – the growth hormone secretagogue receptor type 1a – a G-protein coupled receptor (GPCR) cloned by Howard et al. in 1996. GHS-R1a is expressed in the pituitary gland and hypothalamus and mediates GH release in response to both the endogenous ligand ghrelin and synthetic GHS compounds. Receptor activation triggers intracellular calcium mobilization via Gq/11-mediated phospholipase C signaling.
Can ipamorelin and GHRP-6 be studied with GHRH analogs?
Published research documents synergistic GH secretion when GH secretagogues are co-administered with GHRH analogs in preclinical models – the GHS-R1a and GHRH-R pathways converge at somatotroph exocytosis and potentiate each other. Ipamorelin is frequently selected as the GHS component in these studies when researchers want to isolate GHRH analog activity without cortisol confound from the secretagogue. All co-administration studies involve RUO compounds for in vitro laboratory research only.
Where can I find broader research protocols for GH secretagogue compounds?
The Molecular Edge 2026 Master Index of Peptide Research Protocols provides a comprehensive reference hub for GH secretagogue research frameworks, co-administration study designs, and related compound comparisons across the synthetic peptide research field. For the foundational pharmacological characterization data, referencing the primary literature directly – Raun et al. 1998 for ipamorelin and Bowers et al. 1984 for GHRP-6 – provides the most reliable basis for study design.
Disclaimer: All products sold by Molecular Edge Peptides are strictly intended for laboratory research use only (in vitro). They are not approved for human or animal consumption, or for any form of therapeutic, diagnostic, or clinical use. The information in this article is for educational and scientific reference purposes only. We do not provide usage instructions, dosing guidelines, or any advice regarding personal application of our products. Always consult relevant regulatory frameworks before conducting research with these compounds.
References
- Raun K, Hansen BS, Johansen NL, et al. “Ipamorelin, the first selective growth hormone secretagogue.” Eur J Endocrinol. 1998;139(5):552-561. https://pubmed.ncbi.nlm.nih.gov/9849822/
- Bowers CY, Momany FA, Reynolds GA, Hong A. “On the in vitro and in vivo activity of a new synthetic hexapeptide that acts on the pituitary to specifically release growth hormone.” Endocrinology. 1984;114(5):1537-1545. https://pubmed.ncbi.nlm.nih.gov/6714155/
- Howard AD, Feighner SD, Cully DF, et al. “A receptor in pituitary and hypothalamus that functions in growth hormone release.” Science. 1996;273(5277):974-977. https://pubmed.ncbi.nlm.nih.gov/8688086/
- Bowers CY. “Growth hormone-releasing peptide (GHRP).” Cell Mol Life Sci. 1998;54(12):1316-1329. https://pubmed.ncbi.nlm.nih.gov/9893708/
- Frieboes RM, Murck H, Maier P, Schier T, Holsboer F, Steiger A. “Growth hormone-releasing peptide-6 stimulates sleep, growth hormone, ACTH and cortisol release in normal man.” Neuroendocrinology. 1995;61(5):584-589. https://pubmed.ncbi.nlm.nih.gov/7617137/
- Arvat E, Maccario M, Di Vito L, et al. “Endocrine activities of ghrelin, a natural growth hormone secretagogue (GHS), in humans: comparison and interactions with hexarelin, a nonnatural peptidyl GHS, and GH-releasing hormone.” J Clin Endocrinol Metab. 2001;86(3):1169-1174. https://pubmed.ncbi.nlm.nih.gov/11238504/